|
10 State where in the periodic table these elements appear: (a) (b) (c) (d). This hybridized orbital will repel each other equally and give sp hybridized carbon in ethyne (C 2H 2) its linear shape. Determine the number of valence electrons for the following elements.When the carbon atom is bound to two other atoms with the same shape and energy the hybridization is known to be sp hybridized orbitals.Since carbon forms 2 sigma bonds it will mix 2 of its valence orbitals to form 2 identical orbitals.Carbon in ethyne (C 2H 2) forms 2 sigma bonds and 2 pi bonds.Carbon has four valence (outer) electrons, six protons, and six neutrons. Let us understand it through an example of Ethyne (C 2 H 2 ): This is the sixth element on the periodic table, so we know it has six electrons. The shape of molecules like methane (CH 4), ethane (C 2H 4), ethyne (C 2H 2) is explained in terms of the use of sp 3, sp 2, and sp hybrid orbitals by carbon atoms. When the carbon atom is bound to two other atoms the hybridization is known to be sp.When the carbon atom is bound to three other atoms, the hybridization is sp 2.When carbon, due to its tetravalency, is bonded to four other atoms, it has sp 3 hybridization and tetrahedral arrangement.Hybridization influences the bond length and bond strength in organic compounds. Carbon has 6 electrons out of which 4 valence electrons are present in the 2s2 2p2 outer orbitals of atom. 
Hybridization refers to the concept of combining atomic orbitals in order to form new hybrid orbitals that are appropriate to represent their bonding properties. Element 6 of Periodic table is Carbon with atomic number 6, atomic weight 12.0107. This hybridized orbital will repel each other equally and give sp 3 hybridized carbon in methane (CH 4) its linear shape. When carbon is bonded to four other atoms the hybridization is sp 3 and the arrangement is tetrahedral. And since the carbon in methane (CH 4) has 4 sigma bonds so it will mix 4 of its valence orbitals to form 4 identical orbitals. Hybridization refers to combining atomic orbitals in order to form new hybrid orbitals that are appropriate to represent their properties of bonding. So, this means that there will be 1 shorter and 3 longer bonds in Methane. The nucleus is composed of protons and neutrons. 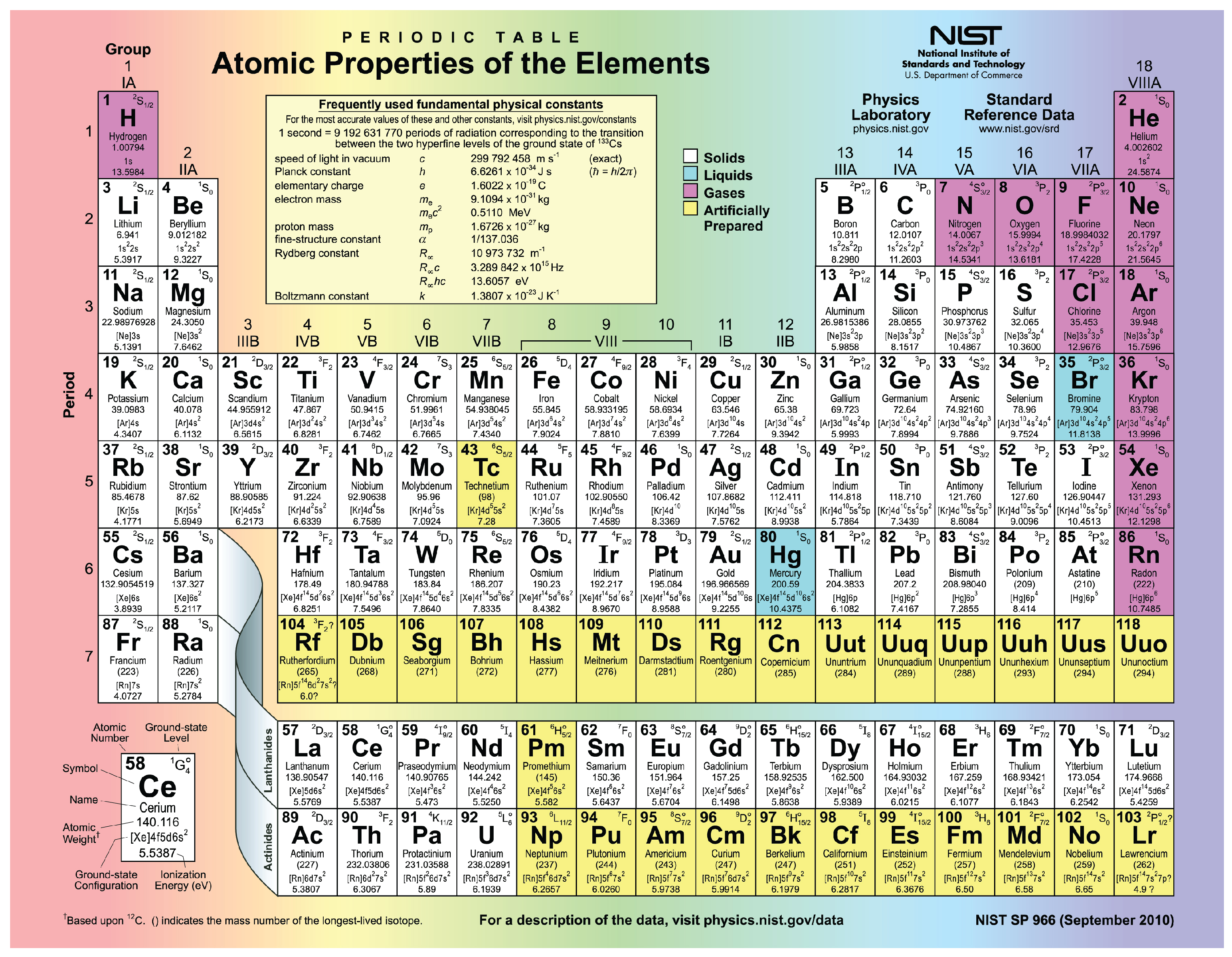
The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
